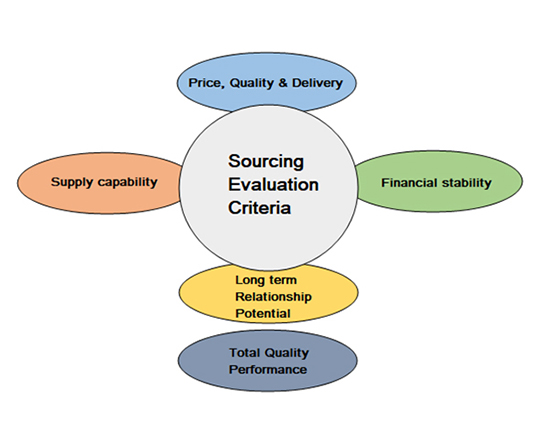
Sourcing Evaluation
At Rushi Pharma, sourcing is not transactional — it is systematic
Manufacturing Site Verification:
- Physical verification of manufacturing facility and coordinates
- Assessment of installed production capacity
- Capacity utilization analysis
- Infrastructure and scale validation
Regulatory & Compliance Review:
- Verification of cGMP systems
- Review of manufacturing licenses and certifications
- Audit history assessment (client / regulatory agency audits)
- Regulatory documentation status
Product Assessment:
- Manufacturing status (regular / campaign / made-to-order)
- In-house specifications and Method of Analysis (MOA)
- Historical price trend evaluation (6–12 months)
- Commercial sustainability analysis
Quality & Technical Validation:
- Technical documentation review
- Pre-shipment samples
- Third-party analysis (if required)
- Ongoing technical coordination
- D-U-N-S: 854321383
- LEI: 9845004484A1C4C82E71
- MSME UDYAM-TS-02-0059615